
Molecular mass is a dimensionless quantity numerically equal to the molar mass. Molecular mass (older name molecular weight) is the mass of a molecule calculated as the sum of the mass of each atom in the molecule multiplied by the number of atoms of that element in the molecule. However, chemists almost always express molar masses in g/mol for convenience. In SI, the unit for molar mass is kg/mol. In other words, it is the mass of one mole of a particular substance. The molar mass is a physical property, which is defined as the mass of a substance divided by its amount of substance in moles. 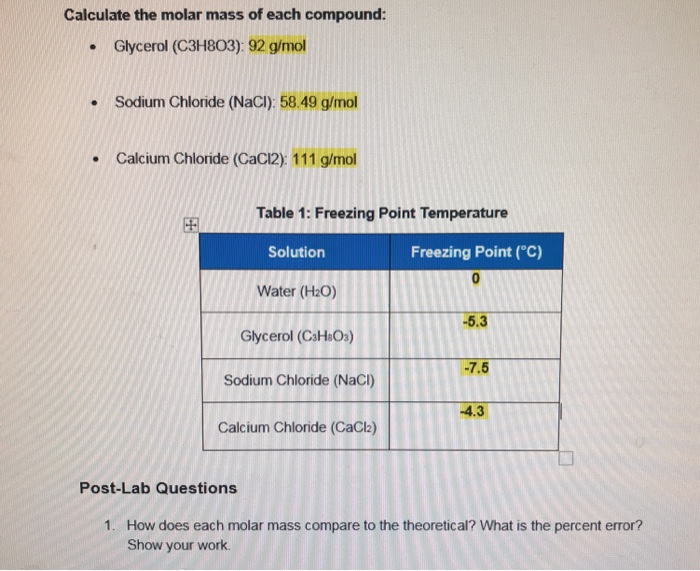
One mole of pure carbon-12 has a mass of exactly 12 grams. The mole as the unit of measurement for the amount of substance is one of the seven base units of the International System of Units (SI). 
In other words, the mole is the amount of substance equal in mass to the combined mass in atomic mass units of the atoms of molecules of the substance multiplied by the Avogadro constant or Avogadro number. An elementary entity may be an atom, a molecule, an ion, an electron, any other particle or specified group of particles.Īvogadro constant N A = 6.02214076×10²³ mol⁻¹ The amount of substance, symbol n, of a system is a measure of the number of specified elementary entities. 
This number is the fixed numerical value of the Avogadro constant, N A, when expressed in the unit mol⁻¹ and is called the Avogadro number. One mole contains exactly 6.02214076×10²³ elementary entities. The mole, symbol mol, is the SI unit of the amount of substance. The mole is used to express the amounts of reactants and products of chemical reactions. In chemistry, it is important to measure their amounts accurately. You can also use our molar mass calculator.All substances consist of atoms or molecules. Basically, you should know how to find the molar masses of any chemical compound now. Of nitrogen) + (six atoms x 16 grams/mole of oxygen) = 189 grams/mole ofįor all other compounds, the general idea is the same. Will be equal to (1 atom x 65 grams/mole of zinc) + (two atoms x 14 grams/mole The brackets multiplied by the subscript two). Multiplied by the subscript two) and six atoms of oxygen (three atoms in Have one atom of zinc, two atoms of nitrogen (one atom inside the brackets (1 atom x 56 grams/mole Fe) + (2 atoms x 35.5 grams/mole of chlorine) = 127 grams/mole of iron (II) chloride For example, in iron (II) chloride, or FeCl 2, you have one atom of iron and two atoms of chlorine. If you have a subscript in a chemical formula, then you multiply the number of atoms of anything next to that subscript by the number of the subscript.įor most compounds, this is easy. (1 atom x 23 grams/mole Na) + (1 atom x 35.5 grams/mole Cl) = 58.5 grams/mole NaClĢ. If we write this as a calculation, it looks like this: If we have a chemical compound like NaCl, the molar mass will be equal to the molar mass of one atom of sodium plus the molar mass of one atom of chlorine. Molar masses of chemical compounds are equal to the sums of the molar masses of all the atoms in one molecule of that compound. To do this, we need to remember a few rules.ġ. How can I find the molar mass of a chemical compound?įor any chemical compound that's not an element, we need to find the molar mass from the chemical formula. Still, aside from the exceptions above, all elements have the same molar mass as the atomic masses on the periodic table. phosphorus is normally found in clumps of four atoms, P 4, and sulfur is found in clumps of eight atoms, or S 8. That each molecule of the element has two atoms of that element stuck together.Īs a result, the formula of hydrogen is H 2, nitrogen is N 2, etc. In the case of hydrogen, nitrogen, oxygen,įluorine, chlorine, bromine, and iodine, the element is diatomic, meaning In some cases, the element is usually found in a differentįorm than just one unbonded atom. So, in our example, carbon has a molar mass of 12.01 grams per mole. For example, if you want to find the molar mass of carbon, you would find the atomic mass of carbon on the periodic table, and this is equal to the molar mass in grams per mole. The molar mass of elements is found by looking at the atomic mass of the element on the periodic table. How can I find the molar mass of an element? It is also sometimes called: Molecular Mass, Molecular Weight, Formula Mass, or Formula Weight. One atomic mass unit (u) is equal to 1/12 the mass of one atom of carbon-12. Molar mass is the mass (in atomic mass units) of one mole of a of a substance.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
